ABOUT CANNABIS-BASED MEDICINES

HISTORY OF CANNABIS-BASED MEDICINES
Medical use of cannabis spans centuries, including widespread therapeutic applications in the 19th century. Recent scientific advances in the endocannabinoid system have transformed it into a modern, research-driven platform for new medicines.
10,000 BCE
Early hemp fibers appear in archaeological records in Central Asia, suggesting cannabis was among the first plants cultivated by humans for practical use.
2700 BCE
Ancient Chinese texts reference cannabis in traditional medicine, documenting its use for pain, inflammation, and other ailments.
1500–1000 BCE
Cannabis appears in Indian religious and medical traditions, including references in early Ayurvedic texts and spiritual ceremonies.
500 BCE
Cannabis spreads through trade routes into the Middle East and Africa, where it is used medicinally, culturally, and agriculturally.
1600s
Cannabis spreads through trade routes into the Middle East and Africa, where it is used medicinally, culturally, and agriculturally.
1800s
Cannabis tinctures are widely available in Western pharmacies and included in medical references for pain relief and sedation.
1906
The Pure Food and Drug Act in the United States begins regulating cannabis-containing medicines, marking the start of modern restriction.
1937
The Marihuana Tax Act effectively criminalizes cannabis in the United States, driven largely by political pressure and misinformation.
1961
The United Nations Single Convention on Narcotic Drugs classifies cannabis as a controlled substance, influencing global prohibition policies.
1970
The U.S. Controlled Substances Act places cannabis in Schedule I, defining it as having no accepted medical use under federal law.
1996
California becomes the first U.S. state to legalize medical cannabis, signaling a major shift in public policy and perception.
2012
Colorado and Washington become the first states to legalize adult-use cannabis, accelerating national reform efforts.
2018
The U.S. Farm Bill legalizes hemp federally, separating hemp from marijuana based on THC content and reopening industrial cannabis markets.
2020s
Cannabis legalization expands globally, with increased focus on plant science, medical research, social equity, and evidence-based policy.

The Endocannibinoid System
What is the endocannabinoid system
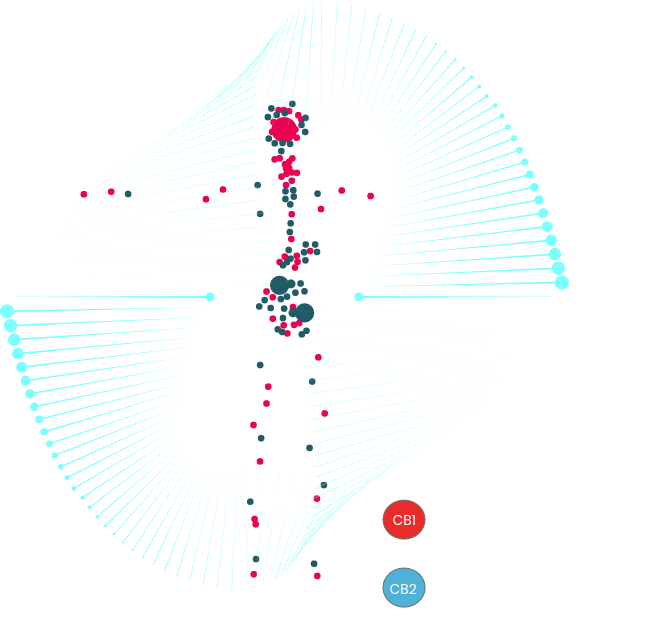
The endocannabinoid system (ECS) is a natural network in the body made up of cannabinoids, receptors, and enzymes that help keep internal functions balanced. These natural compounds are similar to those found in the cannabis plant.
The ECS supports essential processes like mood, sleep, appetite, memory, heart function, and reproduction. When illness or stress disrupts the body’s balance, the ECS helps restore normal functioning and stability.

How does the endocannabinoid system work

Endocannabinoids are not stored within the body; instead, they are produced on demand in response to specific physiological signals. Once synthesized, they move across cell membranes to reach receptor sites, where they bind and are later broken down by specialized metabolic enzymes.
The endocannabinoid system primarily functions as a retrograde signaling network. In this process, activated postsynaptic neurons release endocannabinoids that travel backward across synapses to presynaptic receptors, helping regulate and reduce excessive neurotransmitter release through negative feedback mechanisms.
Endocannabinoids (eCBs) are lipid-based signaling molecules found throughout the body in organs, tissues, and bodily fluids. They serve as key biochemical messengers within the endocannabinoid system (ECS). Although plant-derived cannabinoids such as THC and CBD differ structurally from endocannabinoids, they can interact with the same pathways and produce similar physiological effects, forming the scientific basis of their therapeutic potential.
Major classes of endocannabinoids include N-acylethanolamines (such as anandamide/AEA), N-eicosapentaenoylethanolamine (EPEA), N-docosahexaenoylethanolamine (DHEA), 2-acylglycerols (including 2-AG), and N-acyldopamines (such as NADA)
CB1 receptors (CB1Rs) are primarily located in brain cells and are also present throughout the central nervous system (CNS), peripheral nervous system (PNS), and certain immune system cells.
CB2 receptors (CB2Rs) are widely distributed across the CNS, PNS, and immune system, with particularly high expression in leukocytes.
Transient Receptor Potential (TRP) channels — including TRPA, TRPM, and TRPV — are found across various organs and tissues, especially within brain cells and neurons of the central and peripheral nervous systems.
Peroxisome Proliferator-Activated Receptors (PPARα and PPARγ) are mainly present in adipose tissue and are also expressed in the heart, liver, kidneys, spleen, large intestine, and skeletal muscle.
Biosynthetic Enzymes: These enzymes are responsible for the production of endocannabinoids (eCBs). Key examples include N-Acyl Transferase (NAT), N-Acyl- hosphatidylethanolamine-Hydrolyzing Phospholipase D (NAPE-PLD), Lyso-PLD, Diacylglycerol Lipase alpha and beta (DAGLα/β), and Phospholipase C beta (PLCβ).
Catabolic Enzymes: These enzymes break down endocannabinoids after they have performed their signaling functions. Major examples include Fatty Acid Amide Hydrolase (FAAH), N-Acylethanolamine Acid Hydrolase (NAAH), Cyclooxygenase-2 (COX-2), Lipoxygenase (LOX), and Alpha/Beta Hydrolase Domain enzymes (ABHD).
Transport Proteins: These proteins help move endocannabinoids within cells, across membranes, and between cellular compartments. Notable examples include the FAAH-Like Anandamide Transporter (FLAT), Sterol Carrier Protein-2 (SCP-2), and Fatty Acid Binding Proteins (FABPs).
Chronic pain (musculoskeletal, neuropathic, cancer)

How cannabis-based medicines may help patients

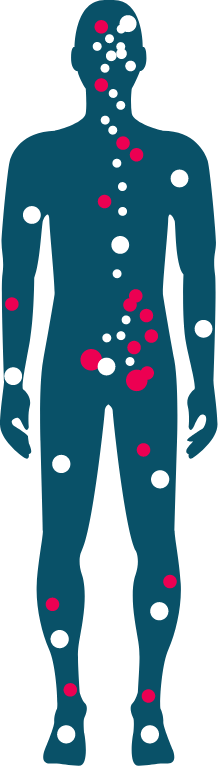
Chronic pain (musculoskeletal, neuropathic, cancer)
Anxiety
Opioid Use Disorder
Multiple Sclerosis (Spasticity)
Rheumatoid Arthritis
Inflammatory Bowel Diseases (Crohn’s)
Autism Spectrum Disorder
Rheumatoid Arthritis
Cancer (glioblastomas, pancreatic)
Eczema / Psoriasis
Sleep
Chemo-induced nausea and vomiting (CINV)


Chronic pain (musculoskeletal, neuropathic, cancer)
Anxiety
Opioid Use Disorder
Multiple Sclerosis (Spasticity)
Rheumatoid Arthritis
Inflammatory Bowel Diseases (Crohn’s)
Autism Spectrum Disorder
Rheumatoid Arthritis
Cancer (glioblastomas, pancreatic)
Eczema / Psoriasis
Sleep
- Sleep
- Anxiety
- PTSD
- Opioid Use Disorder
- Chronic pain (musculoskeletal, neuropathic, cancer)
- Multiple Sclerosis (Spasticity)
- Epilepsy
- Inflammatory Bowel Diseases (Crohn’s)
- Autism Spectrum Disorder
- Rheumatoid Arthritis
- Chemo-Induced Nausea & Vomiting
- Cancer (glioblastomas, pancreatic)
- Eczema/Psoriasis
Chemo-induced nausea and vomiting (CINV)

GLOBAL MARKET
- Core Markets for cannabis-based medicines






- Medical Cannabis Market, Transparency Market Research May 20221
Global market is currently worth over $8bn and is forecast to grow at ~27% CAGR to $53bn by 2027, driven by pain management patients1.

Heartland Herbal Healing Biotech
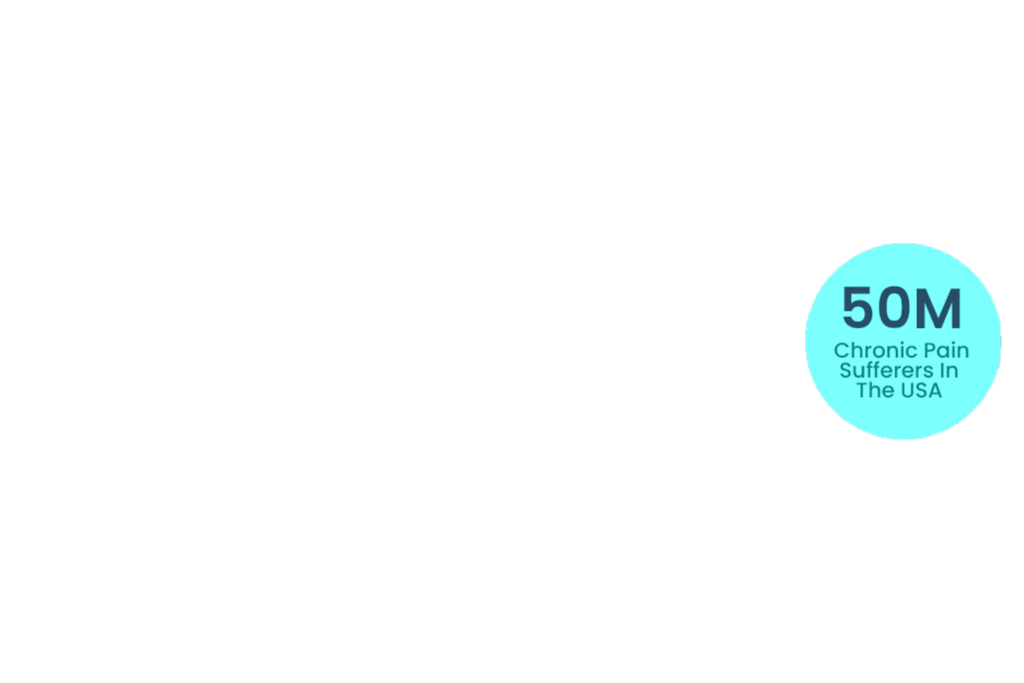
Chronic pain affects a significant portion of the U.S. population, with an estimated over 50 million Americans living with persistent pain and approximately 20 million experiencing high-impact chronic pain that limits daily activities. This burden is expected to rise due to an aging population, increasingly sedentary lifestyles, and growing mental health challenges.
Cannabis-based medicinal products (CBMPs) may offer meaningful societal benefits, including:
- Improved patient outcomes: Traditional opioid therapies are effective for only a small percentage of chronic pain patients and often provide limited long-term relief.
- Reduced adverse effects: The United States continues to face a severe opioid crisis, with tens of thousands of overdose deaths reported annually.
- Lower healthcare and economic costs: Effective alternative therapies could help reduce healthcare utilization, minimize hospitalizations, and support patients in returning to normal daily activities and work.

~50m with chronic pain in the USA


~100m with chronic pain in Europe

Status in the USA
In the United States, cannabis-based medical products (CBMPs) are regulated under a complex framework involving federal restrictions and state-level medical cannabis programs. While cannabis remains classified federally, many U.S. states allow physician-supervised access to medical cannabis for qualifying conditions such as chronic pain, epilepsy, multiple sclerosis–related spasticity, and other serious illnesses. Access is typically provided through state-approved medical cannabis systems rather than traditional FDA drug approval pathways.
These programs operate on a patient-specific basis, reflecting the growing need for alternative therapies where conventional treatments may not provide adequate relief.
Despite rapid market growth, the U.S. medical cannabis sector continues to face several key challenges:
- Limited availability of pharmaceutical-grade, standardized products
- Barriers to consistent patient access and physician adoption
- Low awareness and education regarding clinical benefits and appropriate use.
